Translation, cross-cultural adaptation, and validation of the sino-nasal outcome test (SNOT)-22 for Georgian patients
Abstract
Purpose
The objective of this prospective case-control study was to perform translation, cross-cultural adaptation, and validation of the sino-nasal outcome test 22 (SNOT-22) into the Georgian language.
Methods
The translation and validation of the SNOT -22 questionnaire was performed using the forward-backward translation technique. After proper translation, the translated questionnaire was completed by chronic rhinosinusitis (CRS) patients before and after functional endoscopic sinus surgery (FESS) and by healthy individuals as controls.
Results
SNOT22 was translated into the Georgian language; the pilot study involved 34 patients, the test–retest group consisted of 30 patients with CRS and the control group of 71 patients without CRS complaints; 34 patients were evaluated before surgery and 3 months after surgery. The results showed a good internal correlation with Cronbach’s alpha - 0.88 at the initial examination, and 0.93 at the retest examination; both values suggest good internal consistency within SNOT-22. Pearson’s correlation coefficient was 0.72 (p<0.001), revealing a good correlation between initial scores and retest scores. Our sample of healthy individuals had a median score of 10,11 points and the instrument was able to differentiate between the healthy and the patient group, demonstrating its validity (p<0.0001).
Conclusions
The Georgian version of the SNOT-22 questionnaire is a valid outcome measure for patients with CRS.
Author Contributions
Academic Editor: Raul Isea, Fundación IDEA. Hoyo de la Puerta, Baruta. Venezuela.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2025 Berdia Beridze, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Chronic rhinosinusitis (CRS) is a complex, heterogeneous inflammatory disease affecting 5–12% of the world's population 1. CRS is a chronic condition whose incidence and prevalence are regarded to be increasing in significance. It has an impact on the patient's health-related quality of life (HRQoL). In this regard, it is similar to diabetes and cardiovascular disease 2, 3. Moreover, CRS has a significant impact on the healthcare system. The annual cost per CRS patient is expected to be up to €1501 4. CRS in adults is described as an inflammation of the nose and paranasal sinuses characterised by two or more symptoms lasting more than 12 weeks. One of the symptoms should be either nasal obstruction or nasal discharge, accompanied by facial pain/pressure, a diminished or absent sense of smell, and either endoscopic evidence of nasal polyps or mucopurulent discharge 5.
The World Health Organization (WHO) defines quality of life (QoL) as a person's impression of their situation in life in respect to their goals, expectations, standards, and worries 6. The quality of life can be impacted by a medical disease or its treatment. HRQoL can be diminished by a patient's health and enhanced by adequate medical care.
No population-based epidemiological research regarding CRS have been conducted in Georgia. Several criteria are used by otorhinolaryngologists to assess the severity of symptoms and the efficacy of CRS treatment. In addition to new treatment options for CRS including monoclonal antibodies, it is necessary to have a validated QoL assessment method in order to administer treatment 5.
Due to its reliability, validity, responsiveness, and ease of use, as well as its high dependability for postoperative examination 7, SNOT-22 was chosen to evaluate CRS patients. For the examination of patients with CRS, the questionnaire we picked demonstrated superior to the other 14 QoL surveys. (Table 1). According to Browne et al. The SNOT-22 assesses a vast array of health and HRQoL issues, including physical problems, functional limits, and emotional effects 8.
Table 1. Disease specific tools| RQLQ | SS | F | CST | CSS | RSOM | RSDI | RSI | SN16 | RSUI | SNAQ | SN5 | Col | SN20 | SN22 | |
| Date | 1991 | 1991 | 1993 | 1993 | 1995 | 1995 | 1997 | 1997 | 1998 | 1988 | 2000 | 2001 | 2002 | 1998 | 2000 |
| Items | 28 | 5 | 12 | 12 | 6 | 31 | 30 | 12 | 16 | 10 | 11 | 5 | 7 | 20 | 22 |
| I.C. | 0.94 | 0.78 | 0.78 | 0.73 | 0.89 | 0.40 | 0.70 | 0.90 | 0.91 | ||||||
| R | 0.82 | 1.25 | 0.69 | 0.72 | 1.08 | 0.74 | 0.59 | 0.81 | |||||||
| Scale | 7 | 10 | 4 | 4 | 4 | 6 | 5 | 6 | 5 | 4 | 5 | 7 | 5 | 5 |
The questionnaire comprises 22 symptoms indicative of the patient's health burden 9. Each item measures the intensity of the symptom on a scale ranging from 0 (no problem) to 5 (worst possible symptom), yielding a maximum total questionnaire score of 110. It has been adapted and validated in multiple other languages to date 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23.
However, no specific HRQoL for CRS has been adapted in Georgian. This study's objectives were to translate and adapt the SNOT-22 questionnaire to the Georgian language, test its psychometric qualities, and evaluate the quality of life among the general population and CRS patients before and after treatment.
Materials and Methods
This prospective study was carried out at the Department of Otorhinolaryngology, Tbilisi Medical Academy, New Vision University, Tbilisi, Georgia. The study was approved by the Tbilisi Regional Biomedical Research Ethics Committee (N 07-15-2020/B). Prior to their enrollment in the trial, all patients provided written informed consent. The study only included CRS patients who met the EPOS criteria. Age under 18, pregnancy, and refusal to participate in the study were exclusion factors.
Translation
The translation required five major stages: translation, back-translation, a review by the translation and back-translation committee, an equivalent pre-test with monolingual individuals, and a re-evaluation of the score weights, as proposed by Guillemin 24.
Pilot study
Before the final version of the questionnaire was created, 34 CRS patients were recruited. Each patient independently completed the preliminary translated version of SNOT-22 and discussed the wording and cultural significance of each item with the clinician to ensure that the questionnaire was culturally appropriate. Following the pilot study, a few minor modifications were made to the questionnaire based on patient feedback, resulting in the final version of the Georgian SNOT-22 questionnaire.
Test-Retest study
In a random sample of 55 patients with CRS with or without nasal polyps, test-retest reliability was determined. Patients were asked to complete a questionnaire 14 days apart. The first time was at a typical doctor's visit, and the second time was by letter or a second visit.
Control study
A total of 71 asymptomatic study participants were recruited from hospital personnel and university students. Participants were asked if they suffered from rhinitis or CRS and if they used nose medication. If any of these questions yielded a positive response, the participants were excluded from further study.
Surgery study
In the study group, 34 individuals underwent endonasal surgical therapy. In the "surgical" study group, patients were assessed twice: at baseline and three months after surgery.
Statistical analysis
Internal consistency and test-retest reliability of the Georgian version of the SNOT-22 were examined. Internal consistency describes how pieces within an instrument relate to one another. Using Cronbach's alpha, internal consistency was determined. Estimates of Cronbach's alpha between 0.70 and 0.95 were deemed indicative of acceptable internal consistency 26. Test-retest reliability assesses the consistency of questionnaire responses across a period of time during which symptoms are not anticipated to change. The correlation between the test and retest must be at least 0.70 9.
Using the Mann–Whitney test, the validity of the measurements is determined by the questionnaire's ability to accurately represent differences between previously established groups. We evaluated the questionnaire's ability to produce different scores between the group of patients with CRS with or without nasal polyps and the group of healthy volunteers 23.
The responsiveness of a questionnaire is its ability to identify clinical changes over time. Comparing pre- and post-operative scores using the paired t-test. The size of the effect, which is the mean value of the score variation divided by the standard deviation of the beginning values, can also be used to evaluate responsiveness. By convention, an effect magnitude between 0.2 and 0.5 is considered a moderate increase in quality of life; between 0.5 and 0.8 is a considerable improvement; and greater than 0.8 is a substantial improvement 9.
All statistical analyses were conducted using IBM's SPSS 20 statistical programme. Results with a p-value less than 0.05 were considered statistically significant.
Results
Pilot study
The pilot study included 34 participants, 11 of whom were female (32.4%) and 23 of whom were male (67.6%). Everyone was diagnosed with CRS, with or without nasal polyps. The average age was 48.79 15.20 years old (range 17-80 years). Due to the fact that five patients were unable to locate the area containing the five most significant symptoms, we decided to bold this section of the table and add an arrow to indicate its location.
Test-Retest study
Test-retest reliability was determined in a random sample of 55 CRS patients with or without nasal polyps. The exclusion criteria for the test-retest were a change in treatment and an abrupt change in symptoms related to a common cold or influenza during the interval between the two tests (10 persons). Fifteen participants were removed from the study because they were unavailable for a second review of the questionnaire or because more than fifty percent of the questionnaire slots were unfilled. Thirty individuals were approved for test-retest.
The average age of patients was 50,5 years (range 26-70 years). The subjects consisted of 33.3% females and 66.6% males. The initial mean SNOT-22 score was 56.52 ± 12.24 and the retest score was 58.44 ± 14.40. Cronbach's alpha was 0.88 at the initial assessment and 0.93 at the retest; both numbers indicated that the SNOT-22 had a high degree of internal consistency. Each item's Pearson's correlation analysis resulted in a mean value of 0.72 (p<0.001), indicating a significant correlation. Correspondingly, a high association was found between the initial test scores and the retest scores.
Control Study
Control group included 71 volunteers — 26 female (36.6%) and 45 males (63.4%). The mean age was 45.10 ± 11.37 years, with a range of 30 to 70 years. The control group's mean score was 10.11, with a standard deviation of 8.309. The confidence interval for the mean values was 8.14–12.07 at 95% confidence. Before surgery, we compared the control group to the patient group. The mean SNOT-22 score in the control group was significantly lower than the mean SNOT-22 score in patients with CRS before surgery, which was 61.44 ± 14.24. The tool was able to distinguish across patient groups (p <0.0001), indicating a high level of discriminant validity.
Pre- and Postoperative groups
Three months after surgical treatment, the SNOT-22 scores of 34 CRS patients were compared to their pre-operative ratings for examination of response. The surgical group comprised of 11 female (32.4%) and 23 male (67.6%), with a mean age of 48.79 ± 15.2 years (from 17 years to 80 years). The mean score at the pre-operative evaluation was significantly higher (61.44 ±14.24) than it was three months after surgery (12.32 ± 13.85) (p<0.0001, t = 9.85). (Figure 1). The statistically substantial decrease in post-operative ratings indicated the instrument's responsiveness. Examination of pre-operative scores and scores obtained three months after surgery demonstrated a mean change in scores of 49.12 ± 16.59. The magnitude of the surgery's effect after three months was 1.32, which was considered high (>0.8).
Figure 1.Responsiveness of the Georgian SNOT-22 questionnaire. Comparison of the mean SNOT-22 scores between the preoperative, postoperative and the control groups. Black lines indicate 95% confidence interval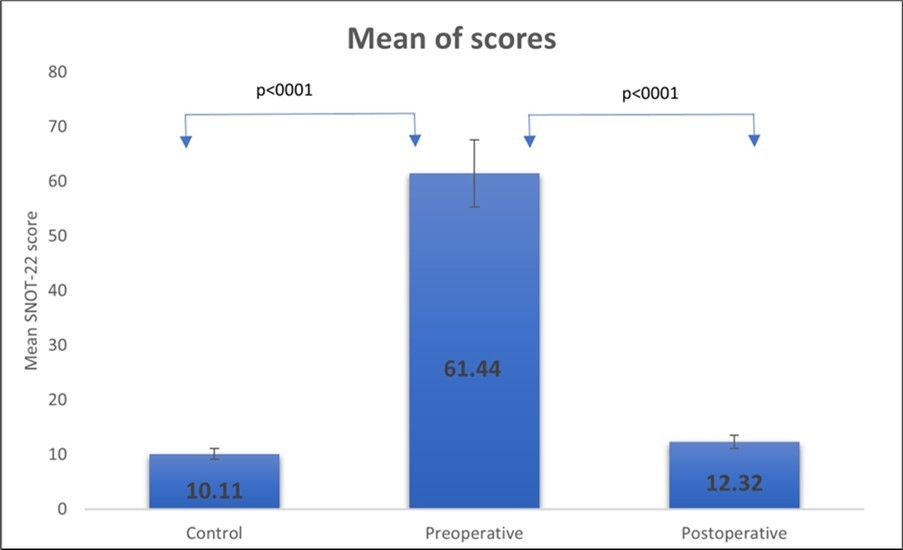
Discussion
We found that the Georgian version of SNOT-22 is a viable outcome-measuring instrument for Georgian patients with sinonasal illness. It exhibits excellent internal consistency, reliability, concurrent validity, and responsiveness.
This questionnaire has already been translated and altered for use in a variety of cultural and language settings. In this work, the SNOT-22 was translated and converted into Georgian using best practises for translation and cultural adaptation 25.
The high usefulness of the SNOT-22 as an assessment tool in patients with chronic rhinosinusitis stems from its capacity to represent the symptom-specific state of the person, highlighting the factors that have the greatest impact on QOL. In addition, the SNOT22 is of utmost importance when surgical therapy is being considered, as it can aid in evaluating the effect of surgery and the likelihood of reaching a realistic outcome.
The questionnaire focuses on a patient's physical, functional, and emotional status; hence, it is essential that it be translated into the patient's native language. Since there was no other standard HRQoL questionnaire translated into Georgian, it was not able to measure the quality of life of CRS patients. We chose to translate, evaluate, and culturally adjust the questionnaire into Georgian so that it may be used in all Georgian ENT clinics. As shown in Table 2, our results were consistent with the original validation by Hopkins et al 9 and comparable to other language translations.
A Cronbach's alpha between 0.70 and 0.95 implies that health surveys have strong internal consistency. According to our data, the Georgian version has a Cronbach's alpha of 0.88, indicating good internal consistency and high reliability of the questionnaire.
Similar to earlier investigations, test-retest reliability was outstanding and indicated proper reliability (Table 2).
Except for Eisenbach et al. 12, our preoperative CRS group exhibited relatively high SNOT-22 scores compared to prior research (Table 2). This may be a result of the Georgian theory that individuals should delay doctor visits as long as possible. The fact that these patients were scheduled for FESS surgery suggests that their condition was likely relatively advanced. After FESS, patients with higher scores report a considerably greater increase in quality of life.
When comparing the mean scores of the questionnaire between the CRS and control groups, as well as between the preoperative and postoperative groups, the Georgian version demonstrated excellent clinical validity (P .0001) and responsiveness (P .0001). These findings support the application of the SNOT-22 scale as a reliable tool for assessing the quality of life (QoL) of patients affected by CRS in the Georgian-speaking population, demonstrating that it can distinguish CRS patients from healthy patients and is sensitive to changes after surgical intervention, as reported by other authors (Table 2).
According to the findings of this study, the Georgian version of the SNOT-22 questionnaire is able to assess changes in HRQoL among surgery patients. We discovered that the difference between the preoperative and postoperative mean SNOT-22 scores was 39.12 points. At three months, the amplitude of the surgical effect was 1.32 and was classed as high (> 0.8). This suggests that FESS is an effective treatment option for the population studied.
Table 2. Georgian ENT clinics| Language | Internal consistency | Test-retest | Validity | Responsiveness | Mean score | |
| CRS | Controls | |||||
| Arabic (11) | 0.803 | 0.907 | <0.001 | <0.001 | 64.2 | 19.5 |
| Brazilian Portuguese (23) | 0.88 | 0.91 | <0.0001 | <0.0001 | 62.4 | 11.4 |
| Czech (19) | 0.9 | 0.86 | NA | NA | 38.5 | 13.7 |
| Danish (20) | 0.83 | 0.7 | NA | NA | 29.7 | NA |
| English (9) | 0.91 | 0.93 | <0.0001 | <0.0001 | 42.0 | 9.3 |
| French (10) | 0.93 | 0.78 | <0.0001 | <0.0001 | 41.0 | 8.3 |
| Georgian | 0.88 | 0.93 | <0.0001 | <0.0001 | 61.44 | 10.11 |
|---|---|---|---|---|---|---|
| German (22) | 0.897 | 0.861 | <0.0001 | <0.0001 | 41.69 | 10.1 |
| German (Austria) (27) | 0.93 | NA | <0.001 | <0.001 | 38.0 | 15.1 |
| Greek (15) | 0.84 | 0.91 | <0.0001 | <0.0001 | 49.6 | 13.0 |
| Hebrew (16) | 0.94 | 0.88 | <0.0001 | <0.001 | 50.4 | 13.2 |
| Italian (13) | 0.86 | 0.85 | <0.008 | <0.001 | 48.9 | 14.3 |
| Lithuanian (17) | 0.89 | 0.72 | <0.0001 | <0.0001 | 52.4 | 16.8 |
| Moroccan (28) | 0.968 | 0.993 | <0.0001 | <0.0001 | 50.4 | 14.5 |
| Russian (12) | 0.816 | 0.98 | <0.0001 | <0.0001 | 67.6 | 9.3 |
| Spanish (18) | 0.91 | 0.87 | <0.0001 | <0.0001 | 47.2 | 4.5 |
| Thai (29) | 0.94 | 0.64 | NA | NA | 38.2 | NA |
| Finnish (31) | 0.879 | 0.93 | <0.0001 | <0.0001 | 46.8 | 8.9 |
| Turkish (30) | 0.88 | 0.97 | <0.0001 | <0.0001 | 64.3 | 15.6 |
Conclusions
For patients with CRS, the Georgian version of the SNOT-22 questionnaire is a valid outcome measure. It has showed strong internal consistency, validity, reproducibility, and responsiveness and is recommended for use with Georgian-speaking CRS patients around the world.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Tbilisi Regional Biomedical Research Ethics Committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Approval NR: N 07-15-2020/B
Statement of Human and Animal rights
All procedures in this study were conducted in accordance with the Tbilisi Regional Biomedical Research CommitteeApproval NR: N 07-15-2020/B
Informed consent
Informed consent was obtained from all individual participants included in the study
Funding
This research received no external funding
Abbreviations
References
- 1.Hastan D, W J Fokkens, Bachert C, R B Newson, Bislimovska J et al. (2011) Chronic rhinosinusitis in Europe--an underestimated disease. A GALEN study. , Allergy 66(9), 1216-1223.
- 2.V K Anand, J D Osguthorpe, Rice D. (1997) Surgical management of adult rhinosinusitis. Otolaryngology--head and neck surgery :. , official journal of American Academy of Otolaryngology-Head and Neck Surgery, 117(3 Pt 2, 50-52.
- 3.R E Gliklich, Metson R. (1995) The health impact of chronic sinusitis in patients seeking otolaryngologic care. Otolaryngology--head and neck surgery :. , official journal of American Academy of Otolaryngology-Head and Neck Surgery 113(1), 104-109.
- 4.E S Lourijsen, W J Fokkens, Reitsma S. (2020) Direct and indirect costs of adult patients with chronic rhinosinusitis with nasal polyps. , Rhinology 58(3), 213-217.
- 5.W J Fokkens, V J Lund, Hopkins C, P W Hellings, Kern R et al. (2020) . European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology, 58(Suppl S29) 1-464.
- 7.A D Morley, H R Sharp. (2006) A review of sinonasal outcome scoring systems - which is best?. Clinical otolaryngology : official journal of ENT-UK ; official journal of Netherlands Society for Oto-Rhino-Laryngology &. , Cervico-Facial Surgery 31(2), 103-109.
- 8.J P Browne, Hopkins C, Slack R, S J Cano. (2007) The Sino-Nasal Outcome Test (SNOT): can we make it more clinically meaningful?. Otolaryngology--head and neck surgery : official journal of. , American Academy of Otolaryngology-Head and Neck Surgery 136(5), 736-741.
- 9.Hopkins C, Gillett S, Slack R, V J Lund, J P Browne. (2009) Psychometric validity of the 22-item Sinonasal Outcome Test. Clinical otolaryngology : official journal of ENT-UK ; official journal of Netherlands Society for Oto-Rhino-Laryngology &. , Cervico-Facial Surgery 34(5), 447-454.
- 10.C de Dorlodot, Horoi M, Lefebvre P, Collet S, Bertrand B et al. (2015) French adaptation and validation of the sino-nasal outcome test-22: a prospective cohort study on quality of life among 422 subjects. Clinical otolaryngology : official journal of ENT-UK ; official journal of Netherlands Society for Oto-Rhino-Laryngology &. , Cervico-Facial Surgery 40(1), 29-35.
- 11.Asiri M, Alokby G. (2019) Validation and Cross-cultural Adaptation of the Sinonasal Outcome Test (SNOT)-22 for the Arabian Patient Population. , Cureus 11(4), 4447-10.
- 12.Eisenbach N, Matot S, Nemet A, Sela E, Marshak T et al. (2020) Sino-nasal outcome test-22: Cross-cultural adaptation and validation in Russian speaking patients. Clinicalotolaryngology : official journal of ENT-UK ; official journal of Netherlands Society for Oto-Rhino-Laryngology &. , Cervico-Facial Surgery 45(3), 350-356.
- 13.Mozzanica F, Preti A, Gera R, Gallo S, Bulgheroni C et al. (2017) Cross-cultural adaptation and validation of the SNOT-22 into Italian. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 274(2), 887-895.
- 14.G P Caminha, Junior Melo, T J, Hopkins C, Pizzichini E et al. (2012) SNOT-22: psychometric properties and cross-cultural adaptation into the Portuguese language spoken in Brazil. Brazilian journal of otorhinolaryngology. 78(6), 34-39.
- 15.V A Lachanas, Tsea M, Tsiouvaka S, J K Hajiioannou, C E Skoulakis et al. (2014) The sino-nasal outcome test (SNOT)-22: validation for Greek patients. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 271(10), 2723-2728.
- 16.Galitz Shapira, Halperin Y, Bavnik D, Y, Warman M. (2016) Sino-Nasal Outcome Test-22: Translation, Cross-cultural Adaptation, and Validation in Hebrew-Speaking Patients.Otolaryngology--head and necksurgery :official journal of. , American Academy of Otolaryngology-Head and Neck Surgery 154(5), 951-956.
- 17.Vaitkus S, Padervinskis E, Balsevicius T, Siupsinskiene N, Staikuniene J et al. (2013) Translation, cross-cultural adaptation, and validation of the sino-nasal outcome test (SNOT)-22 for Lithuanian patients. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 270(6), 1843-1848.
- 18.de los Santos, Reyes G, del Castillo P, Fragola R, C et al. (2015) Cross-cultural adaptation and validation of the sino-nasal outcome test (SNOT-22) for Spanish-speaking patients. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 272(11), 3335-3340.
- 19.Schalek P, Otruba L, Hahn A. (2010) Quality of life in patients with chronic rhinosinusitis: a validation of the Czech version of SNOT-22 questionnaire. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 267(3), 473-475.
- 20.Lange B, Thilsing T, Al-kalemji A, Baelum J, Martinussen T et al. (2011) . validated for Danish patients.Danish medical bulletin, 58(2), A4235 , The Sino-Nasal Outcome Test 22.
- 21.Jalessi M, Farhadi M, S K Kamrava, Amintehran E, Asghari A et al. (2013) The reliability and validity of the persian version of sinonasal outcome test 22 (snot 22) questionnaires.Iranian Red Crescent medical journal. 15(5), 404-408.
- 22.Albrecht T, A G Beule, Hildenbrand T, Gerstacker K, Praetorius M et al. (2022) Cross-cultural adaptation and validation of the 22-item sinonasal outcome test (SNOT-22) in German-speaking patients: a prospective, multicenter cohort study. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 279(5), 2433-2439.
- 23.E M Kosugi, V G Chen, V M Fonseca, M, Neto Mendes et al. (2011) Translation, cross-cultural adaptation and validation of SinoNasal Outcome Test (SNOT): 22 to Brazilian Portuguese. Brazilian journal of otorhinolaryngology. 77(5), 663-669.
- 24.D L Streiner. (2003) Starting at the beginning: an introduction to coefficient alpha and internal consistency.Journal of personality assessment. 80(1), 99-103.
- 25.Wild D, Grove A, Martin M, Eremenco S, McElroy S et al. (2005) ISPOR Task Force for Translation and Cultural Adaptation. Principles of Good Practice for the Translation and Cultural Adaptation Process for Patient-Reported Outcomes (PRO) Measures: report of the ISPOR Task Force for Translation and Cultural Adaptation. Value in health : the journal of the International Society for Pharmacoeconomics and Outcomes Research 8(2), 94-104.
- 26.C B Terwee, S D Bot, Boer M R de, Windt D A van der, D L Knol et al. (2007) Quality criteria were proposed for measurement properties of health status questionnaires.Journal of clinical epidemiology. 60(1), 34-42.
- 27.Riedl D, Dejaco D, T B Steinbichler, Innerhofer V, Gottfried T et al. (2021) Assessment of health-related quality-of-life in patients with chronic Rhinosinusitis - Validation of the German Sino-Nasal Outcome Test-22 (German-SNOT-22).Journal of psychosomatic research. 140-110316.
- 28.Adouly T, Adnane C, Khallouk A, Chenguir M, Rouadi S et al. (2017) Moroccan adaptation and validation of the rhinosinusitis quality-of-life survey. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology- Head and Neck Surgery. 274(3), 1507-1513.
- 29.Numthavaj P, Bhongmakapat T, Roongpuwabaht B, Ingsathit A, Thakkinstian A. (2017) The validity and reliability of Thai Sinonasal Outcome Test-22. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 274(1), 289-295.
- 30.Cetin Cakir, Kumus A, Keskinoglu O, Sutay P, S et al. (2019) Turkish validation of the Sino-Nasal Outcome Test-22. Clinicalotolaryngology :official journal of ENT-UK ;official journal of Netherlands Society for Oto-Rhino-Laryngology &Cervico-Facial Surgery. 44(4), 557-564.
- 31.Koskinen A, Hammarén-Malmi S, Myller J, Mäkelä M, Penttilä E et al. (2021) Translation, cross-cultural adaptation, and validation of the sino-nasal outcome test (snot)-22 for Finnish patients. European archives ofoto-rhino-laryngology :official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) :affiliated with the German Society for Oto-Rhino-Laryngology -Head and Neck Surgery. 278(2), 405-410.
